Paediatric haemodialysis

6008 CAREsystem Paed therapy system: solutions for paediatric patients
- Targeting growth and cardiovascular health
- Makes highly efficient HDF therapy accessible to children
- Proven system works together, aiming for the best possible safety

Haemodialysis: targeting growth and cardiovascular health
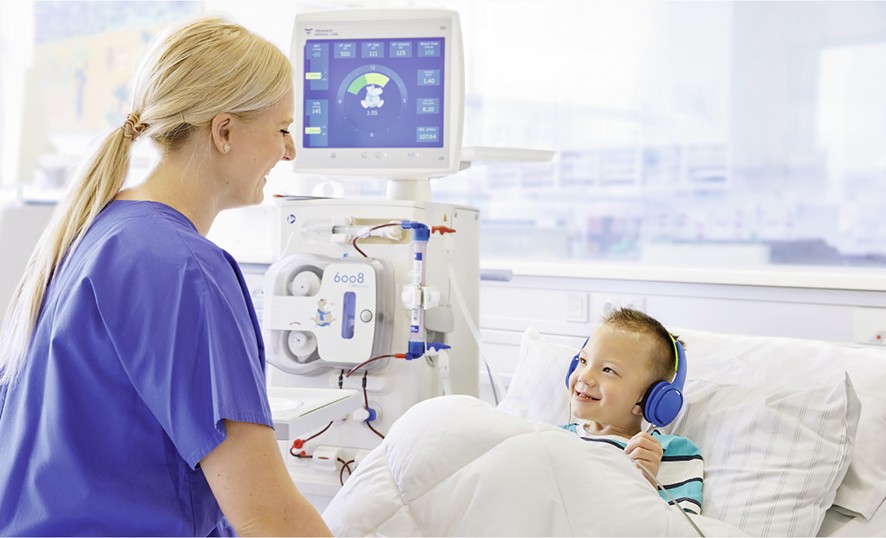
Haemodialysis (HD) is also a treatment option for children with Chronic Kidney Disease (CKD). Older children and adolescents eventually prefer HD to Peritoneal Dialysis (PD) because it affects less their body image than if they were to have a PD catheter.
Children with kidney disease are committed to a lifetime of renal replacement therapy and as many of the cardiovascular effects of dialysis are cumulative, optimisation of dialysis therapy in children is very important.
The 6008 IFU states the low volume option allows treatment of patients with a dry weight of between 10-40kg.
Reduced complexity in paediatric therapy: the 6008 CAREsystem Paed
With the 6008 CAREsystem Paed, Fresenius Medical Care offers a haemodialysis system specially designed for the treatment of paediatric patients. It fulfils the highest quality standards for the best treatment outcomes, making HDF therapy available to children and easy to perform.
- Approved and validated machine for paediatric therapies
- Sophisticated safety concept: adapted software, including additional safety features to monitor the venous path (VAM, VenAcc)
- Enables HighVolumeHDF®, an advanced method aiming at maximising substitution volumes in HDF also at low blood flows
6008 CAREset: suitable for paediatric treatments
- Minimised volume of the extracorporeal circuit*
- Air-free design
- Reduced burden of potentially hazardous substances
- Free of phthalates like DEHP
- Less PVC (22% less than the 5008 AV-Set)
- Less plasticisers in total due to Biofine components (plasticiser-free)
6008 Paed: a well thought-out system
- Software adapted to paediatric needs for improved safety
- E.g. adapted air/microbubble detection sensitivity and blood flow rates
- High balancing accuracy
- User set-up designed for paediatric treatments
- Increased probability of early detection of venous needle dislodgement (VAM–Venous Access Monitor, VenAcc)
* Extracorporal volume: 105 ml with the 6008 CAREset
5008S CorDiax Paediatric Haemodiafiltration (HDF): the recommended course of action
There is growing evidence that Haemodiafiltration (HDF) is an effective way to prevent cardiovascular disease in children.
The 5008S CorDiax Paed machine makes highly efficient HDF therapy also accessible for children.
With 5008S CorDiax Paed, Fresenius Medical Care offers a proven system to treat young patients starting from 10 kg* of dry weight. All components of the system, dialysis machine, blood line and the dialysers have been tested to perfectly work together as an entity.
With our 5008S CorDiax Paed therapy system, we are offering solutions for the specific challenges associated to HD in paediatric patients:
- Management of low volumes
- Best therapy outcome
- Well-being during treatment
- Additional safety features
- Best handling and smart accessories
* As of software version 4.63.
Managing low volumes
- Sensitive machine settings and limits
- Blood flow is limited according to body weight
- Reduced bolus rate according to blood flow
- Reduced blood flow of 50 ml/min for a gentle start of dialysis
- Adapted air and microbubble detection sensitivity
- Paediatric user setup
- Paediatric AV blood line set with reduced fill volume of 114 ml
The 5008S CorDiax Paed software allows therapeutic parameters and thresholds to be adjusted within paediatric ranges, for example:
- A re-infusion volume to be chosen within the range of 60-480 ml
- An ultrafiltration rate to be limited to max. 1500 ml/h
- A heparin rate to be set within the range of 0.5-10 ml/h
Best therapy outcome
Targeting best therapy outcomes with the 5008S Paed
- HighVolumeHDF with AutoSubplus and FX CorDiax aims at maximising substitution volume, also at low blood flows, to achieve clearance goals
Dialysers with reduced volume
| Special dialysers | FX paed | FX CorDiax 40 |
|---|---|---|
| Effective surface area | 0.2 m² | 0.6 m² |
| Blood priming volume | 18 ml | 32 ml |
| Recommended blood flow range | 30-100 ml/min | 50-200 ml/min |
| Membrane | Helixone® | Helixone® plus |
| Application | HD | HD/HF/HDF |
| Sterilisation method | INLINE steam |
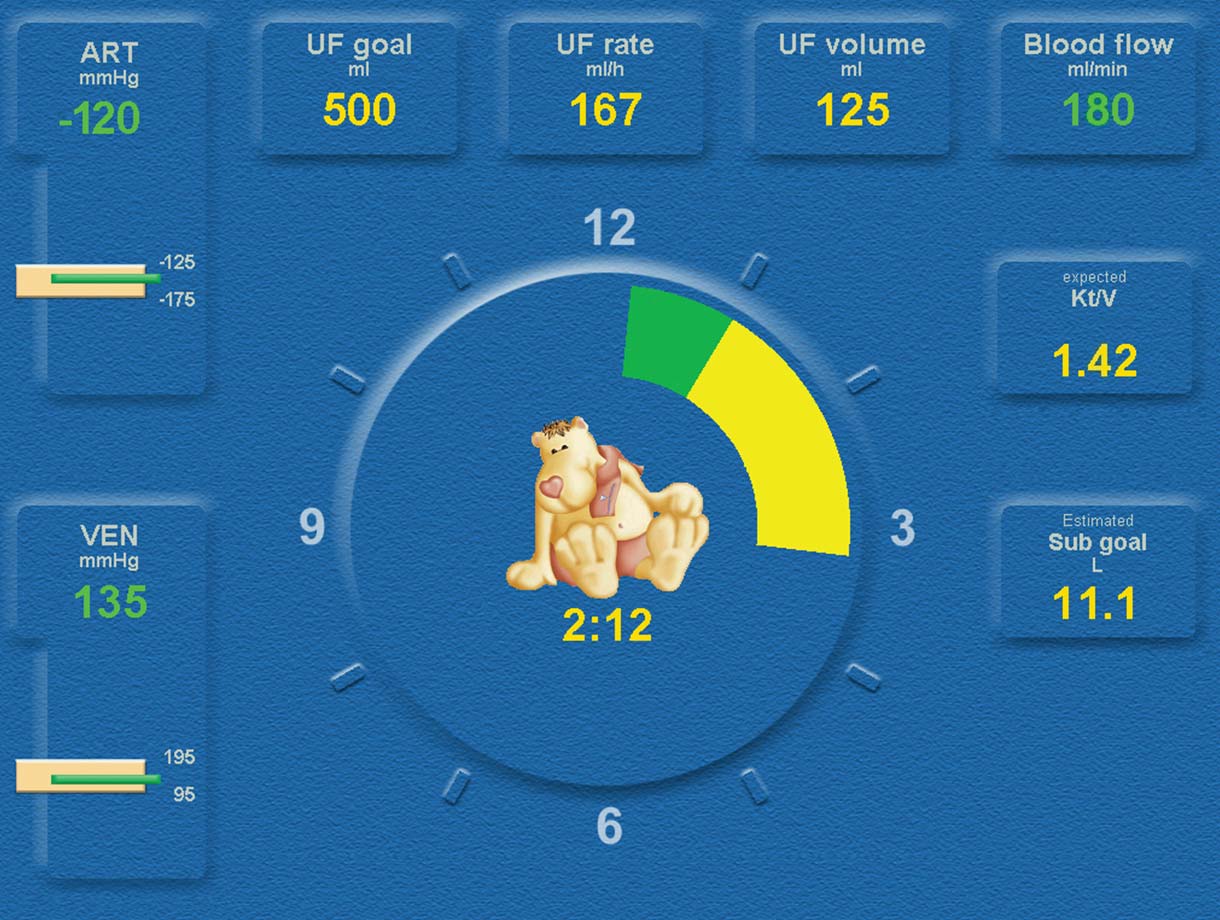
Blood Volume Monitor
Well-being during treatment
- The BVM-Blood Volume Monitor helps to maintain haemodynamic stability during treatment
- Preset inflation pressure of BPM cuff for painless blood pressure measurements
Additional safety features
- Higher probability of early detection of venous needle dislocation with the VAM–Venous Access Monitor (standard)
- The additional safety feature, VenAcc enables detection of venous dislodgment (option)
- Nonhazardous phtalate-free bloodlines
Best handling and clever accessories adapted to paediatric use
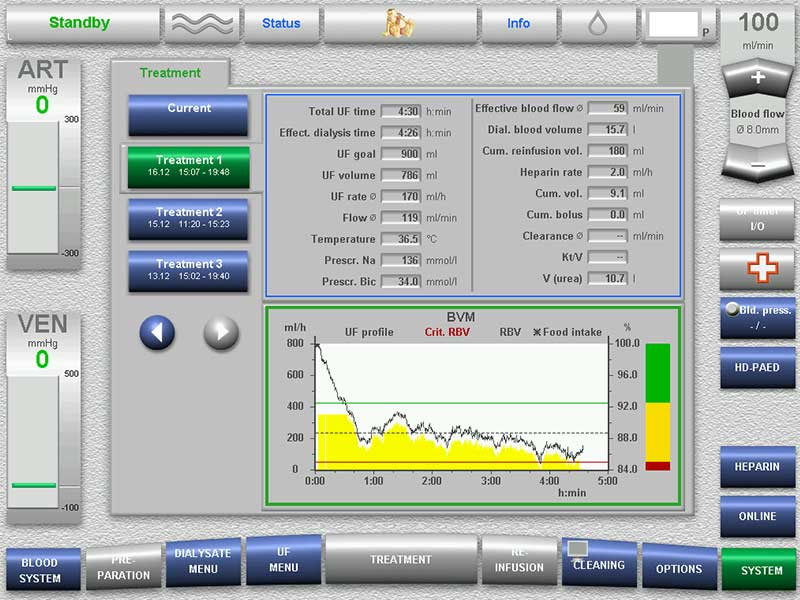
The 5008S CorDiax therapy system is known for its intuitive user guidance philosophy:
- Centralised operation and information via a spacious touch screen
- Simple and logical data entry
- Sophisticated, stress-free handling of alarms during treatment
- Easier in-house training with the new mode for the simulation of blood detection
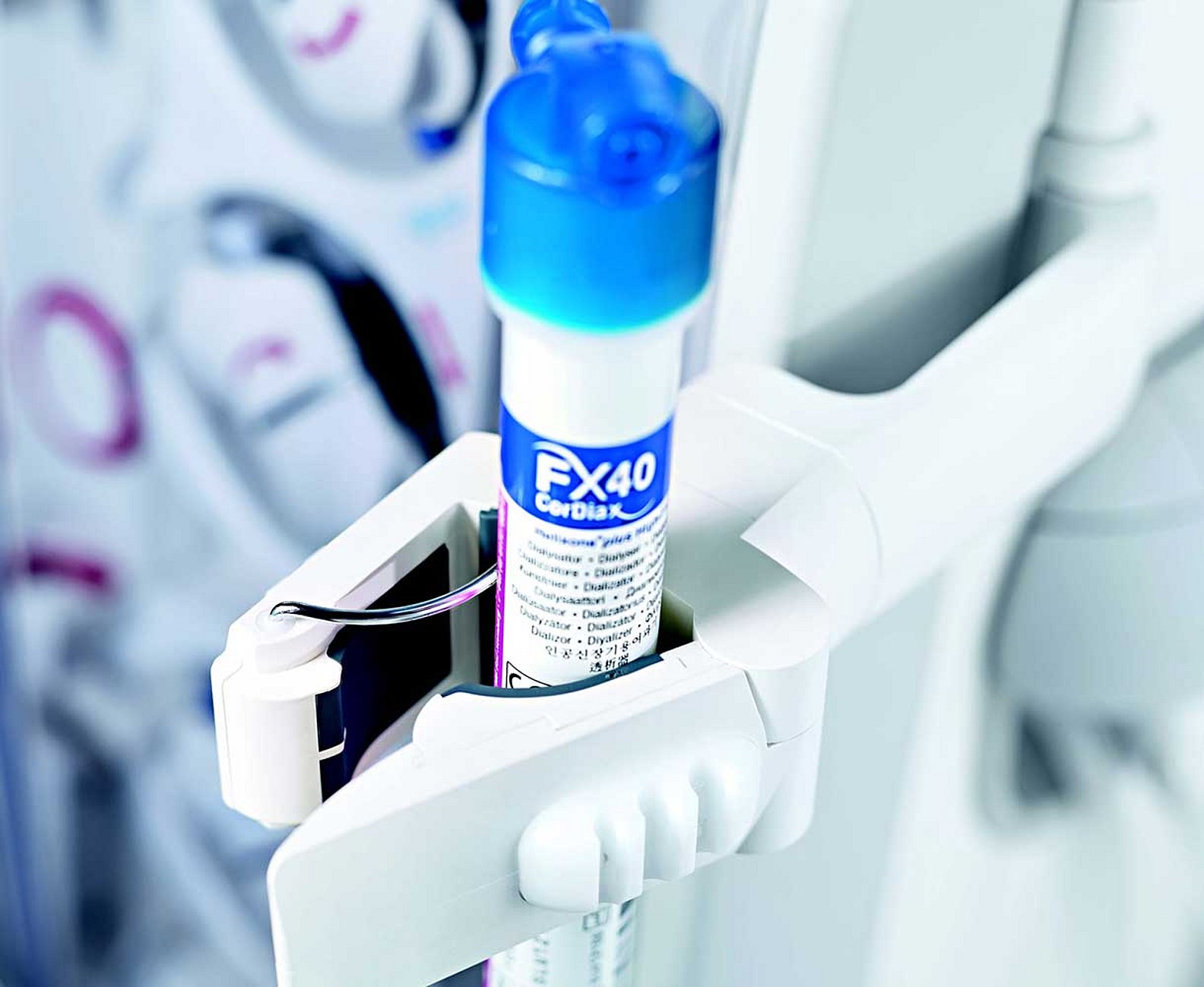
CorDiax filter small dialyser holder

BPM Blood Pressure Measurement cuff for children with an arm circumference of 13-20 cm
If you would like to order this product via the NHS Supply Chain Catalogue, please visit the following link: NHS Supply Chain Online Catalogue
Additional information relating to multiBic or Calrecia can be found in the critical care section of our product information page.
Adverse Events Reporting
Adverse events should be reported. Reporting forms and information can be found at
https://yellowcard.mhra.gov.uk/ or search for MHRA Yellowcard in the Google Play or Apple app store. Adverse events should also be reported to Fresenius Medical Care on 01623 445100.
UK/HEMA/FME/0922/0005 Date of Preparation: November 2022